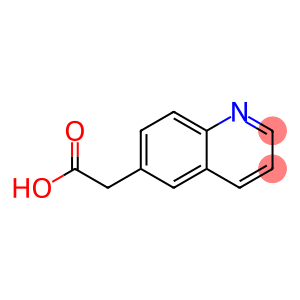
2-(quinolin-6-yl)acetic acid
6-Quinoline acetic acid
CAS: 5622-34-4
Molecular Formula: C11H9NO2
2-(quinolin-6-yl)acetic acid - Names and Identifiers
| Name | 6-Quinoline acetic acid |
| Synonyms | OK-J-05024 6-QuinolineAceticAci 6-Quinolylacetic acid 6-Quinolineacetic Acid 6-Quinoline acetic acid quinolin-6-ylacetic acid Quinolin-6-Yl-Acetic Acid 2-(quinolin-6-yl)acetic acid 6-Quinolylacetic acid 2-(Quinolin-6-yl)acetic acid |
| CAS | 5622-34-4 |
| InChI | InChI=1/C11H9NO2/c13-11(14)7-8-3-4-10-9(6-8)2-1-5-12-10/h1-6H,7H2,(H,13,14) |
2-(quinolin-6-yl)acetic acid - Physico-chemical Properties
| Molecular Formula | C11H9NO2 |
| Molar Mass | 187.19 |
| Density | 1.297±0.06 g/cm3(Predicted) |
| Melting Point | 225 °C (decomp) |
| Boling Point | 388.8±17.0 °C(Predicted) |
| Flash Point | 188.915°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 3.65±0.30(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.661 |
| MDL | MFCD07776794 |
2-(quinolin-6-yl)acetic acid - Introduction
Quinoline acid is an organic compound whose chemical structure contains a quinoline ring and an acetic acid group. The following is an introduction to the nature, use, formulation and safety information of 6-Quinoline acetic acid:
Nature:
-Appearance: 6-Quinoline acetic acid is white to light yellow solid powder.
-Solubility: It is soluble in alcohol, chloroform and dimethylformamide, slightly soluble in water.
-Melting point: Its melting point is about 156-160 degrees Celsius.
Use:
6-Quinoline acid has many uses in organic synthesis:
-It can be used as a catalyst for the synthesis of asymmetric carbazole and isoxazole compounds.
-It can also be used to synthesize other organic compounds containing quinoline ring, such as phosphoramide drugs.
-Other applications include optical materials and coordination chemistry.
Preparation Method:
The preparation method of 6-Quinoline acetic acid generally involves the following steps:
-First, an acetaldehyde-containing diol and a para-substituted aniline are condensed under acid catalysis to generate a para-substituted quinoline compound.
-Then, the quinoline compound is reacted with acetic anhydride under alkaline conditions to generate 6-Quinoline acetic acid.
Safety Information:
There is limited data on the safety of Quinoline acid. But in general, it may have a certain irritation and sensitization. Therefore, in handling and use, appropriate safety measures should be taken, such as wearing protective gloves, glasses and laboratory coats to avoid skin contact and inhalation. In addition, it should be stored in a dry, cool place, away from fire and flammable substances. It is best to use in a well-ventilated place, and pay attention to the fire and explosion hazards during storage and handling. If inhaled or exposed to the substance, wash the affected area immediately and seek medical help if necessary.
Nature:
-Appearance: 6-Quinoline acetic acid is white to light yellow solid powder.
-Solubility: It is soluble in alcohol, chloroform and dimethylformamide, slightly soluble in water.
-Melting point: Its melting point is about 156-160 degrees Celsius.
Use:
6-Quinoline acid has many uses in organic synthesis:
-It can be used as a catalyst for the synthesis of asymmetric carbazole and isoxazole compounds.
-It can also be used to synthesize other organic compounds containing quinoline ring, such as phosphoramide drugs.
-Other applications include optical materials and coordination chemistry.
Preparation Method:
The preparation method of 6-Quinoline acetic acid generally involves the following steps:
-First, an acetaldehyde-containing diol and a para-substituted aniline are condensed under acid catalysis to generate a para-substituted quinoline compound.
-Then, the quinoline compound is reacted with acetic anhydride under alkaline conditions to generate 6-Quinoline acetic acid.
Safety Information:
There is limited data on the safety of Quinoline acid. But in general, it may have a certain irritation and sensitization. Therefore, in handling and use, appropriate safety measures should be taken, such as wearing protective gloves, glasses and laboratory coats to avoid skin contact and inhalation. In addition, it should be stored in a dry, cool place, away from fire and flammable substances. It is best to use in a well-ventilated place, and pay attention to the fire and explosion hazards during storage and handling. If inhaled or exposed to the substance, wash the affected area immediately and seek medical help if necessary.
Last Update:2024-04-09 21:32:42
Supplier List
Spot supply
Product Name: 2-(Quinolin-6-yl)acetic acid Visit Supplier Webpage Request for quotationCAS: 5622-34-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6-Quinoline acetic acid Request for quotation
CAS: 5622-34-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 5622-34-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 6-QUINOLINEACETIC ACID Request for quotation
CAS: 5622-34-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5622-34-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 6-Quinolineacetic Acid Visit Supplier Webpage Request for quotation
CAS: 5622-34-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5622-34-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-(Quinolin-6-yl)acetic acid Visit Supplier Webpage Request for quotationCAS: 5622-34-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6-Quinoline acetic acid Request for quotation
CAS: 5622-34-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 5622-34-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 6-QUINOLINEACETIC ACID Request for quotation
CAS: 5622-34-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5622-34-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 6-Quinolineacetic Acid Visit Supplier Webpage Request for quotation
CAS: 5622-34-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5622-34-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



